Tamiflu doubts: answers from the BMJ
Updated on 08 December 2009
Deborah Cohen from the BMJ answers questions raised by the report she worked on, which has cast new doubts on the effectiveness of the antiviral drug Tamiflu.
The unseen data
So why does it matter that eight of the 10 clinical trials analysed by Professor Kaiser were never fully published?
Why should the Cochrane team expect to get access to commercially sensitive company-owned data?
The cost
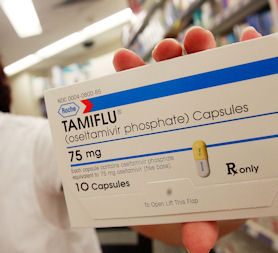
It is estimated the UK government has spent £500m on Tamiflu. Could the money have been better spent in other areas?
US v Europe
Why does the US Food and Drug Administration (FDA) not recognise claims that Tamiflu reduces complications, when Roche says it does in Europe?
What next?
Will the BMJ be pressing for any change in policy from the government as a result of this study?
The rest
Some people might now worry about Tamiflu's effectiveness in other areas - such as for young children and pregnant women. What would you say to reassure them?